
Fat. What a terrible word. It’s what we don’t want. Actually we need a fast way to get rid of it.
Or so the word fat is generally perceived. But that’s wrong. We need to eat fat, and we need fat in our bodies (just not too much of it). People with a genetic condition that means they can’t store fat normally are not healthy.
Fats – or lipids as scientists call this group of materials – are simply substances that won’t dissolve in water. There are many types. But when we think of fat in food, or in our bodies, we are usually thinking of a group of substances whose basic unit is called a fatty acid (acid because it has some acidic properties). Each molecule of a fatty acid is made of a chain of carbon atoms, joined one to another, with hydrogen atoms filling all the spare bonds either side: but at one of the chain is the ‘acid’ group, also called a carboxyl group, and usually shown in chemical structures as -COOH. Most fatty acids common in the kitchen – and in our bodies – have 16 or 18 carbon atoms in the chain. (They have an even number because they are built up by adding consecutively molecules with two carbon atoms.)
But, to make things a little more complex, along the chain of carbon atoms, there may be links called ‘double bonds’ where the spare bonds are not filled with hydrogen atoms, and the adjacent carbon atoms join with two bonds rather than one. An important consequence is that this introduces a kink into the molecule. The figure shows the shapes of the molecules of two common fatty acids, each with a chain of 18 carbon atoms, one without any double bonds (this is called a saturated fatty acid) and the other with one double bond (this is called a mono-unsaturated fatty acid): these are called respectively stearic acid and oleic acid and they are major fatty acids in meat fat and in olive oil respectively. Some fatty acids have more than one double bond in the molecule (they are called polyunsaturated): for instance, linoleic acid, a major fatty acid in sunflower oil, has 18 carbon atoms and two double bonds – and their shape is very ‘kinky’.
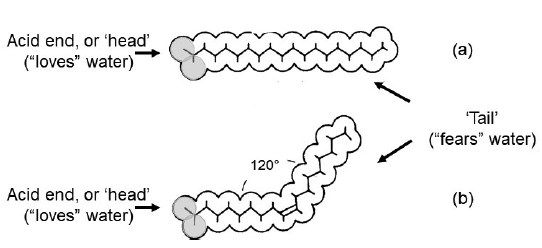
The figure also shows that the ‘acid’ end of the fatty acid ‘loves’ water whereas most of the molecule – the ‘fatty’ bit – does not. Nature (evolution) has found a way of making the molecules entirely ‘fatty’. The acid groups of three fatty acids may link to a small alcohol-related molecule called glycerol (or glycerine). Then we have a ‘triglyceride’ or more properly a triacylglycerol (TG). TGs are what we usually think of as fat. Solid fats like beef suet or butter, liquid fats like sunflower oil and olive oil, and indeed the semi-solid fat that we can feel in the tissues just under our skin – these are all TGs. The fatty acids in TGs may be saturated or unsaturated. In meat fat many are saturated and, because of the straight nature of the molecules, they can pack together easily (something like a crystal) and are mostly solid at room temperature. The more unsaturated the fatty acids, the more liquid the fat. Olive oil (mainly oleic acid, one double bond) is liquid in the kitchen but goes solid in the fridge. Sunflower oil, with mostly polyunsaturated fatty acids, is liquid even in the fridge.
In a later blog we will look more at the metabolic pathways by which these fats can be stored in our own fat stores, and how they can be ‘mobilised’ when we need the stored energy (or when we want to shed a bit of weight).

Title: Understanding Human Metabolism
Author: Keith Frayn
Paperback ISBN: 9781009108522
Latest Comments
Have your say!